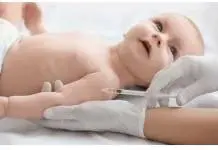
The FDA has approved the use of Apretude for preventing HIV infection – the first injectable medication for the sexually-transmitted disease. Apretude will be recommended for people who weigh at least 77 pounds and test negative for HIV immediately before the need for the drug.
Apretude will be an injectable alternative to people who find it difficult to keep up with oral HIV-prevention drugs such as Truvada which must be taken every day. Apretude injection is taken twice – at one-month intervals – and then followed by one shot every two months.
Two different studies conducted on Apretude in 13 countries indicated that the injectable is more effective at preventing HIV than Truvade oral drug. In the first study where almost 5,000 participants were recruited – including cisgender men and transgender women – who were administered with the injection after taking the oral drug cabotegravir were found to be 69% at lesser risk of getting HIV infection.
For the second study involving over 3,200 cisgender women at risk of HIV infection, participants who got Apretude and took cabotegravir were found to be 90% lesser at risk of getting HIV disease than for people who received Truvada. Debra Birnkrant, director of the Division of Antivirals in the FDA’s Center for Drug Evaluation and Research, said Apretude injection is more effective and easier to use for HIV prevention than daily pills.
The FDA said gays, drug addicts, and people suffering from depression and poverty usually find it difficult to take required drugs on a daily basis, but they would find Apretude injection more helpful for their overall health. According to the CDC, gays – men who have sex with fellow men – occasioned 66% of new HIV cases in 2019.
US health officials said a fourth of the 1.2 million people who were qualified to get HIV-prevention drugs got them in 2020, even though only 3% of eligible people got them in 2015. Every year, about 1.7 new HIV infections are recorded in the United States, and the FDA advised people who get infected while taking Apretude to immediately switch to an appropriate HIV-treatment course.
ViiV Healthcare – whose majority stake is controlled by GlaxoSmithKline – will begin to distribute Apretude in early 2022. The drug is currently approved only in the US but may move to other major markets in the coming years.